Tirzepatide’s Approval
The weight-loss drug tirzepatide has gained approval from the US Food and Drug Administration (FDA) and the UK’s Medicine’s and Healthcare products Regulatory Agency for adults who are overweight or have obesity and at least one weight-related condition.
The drug has shown exceptional weight reduction in clinical trials, leading to its approval for weight management.
Unprecedented Weight Loss
In clinical trials, tirzepatide demonstrated an average weight reduction of almost 21%, marking it as a breakthrough medication for weight loss. This level of weight loss is comparable to that achieved through surgery.
Zepbound: A New Hope
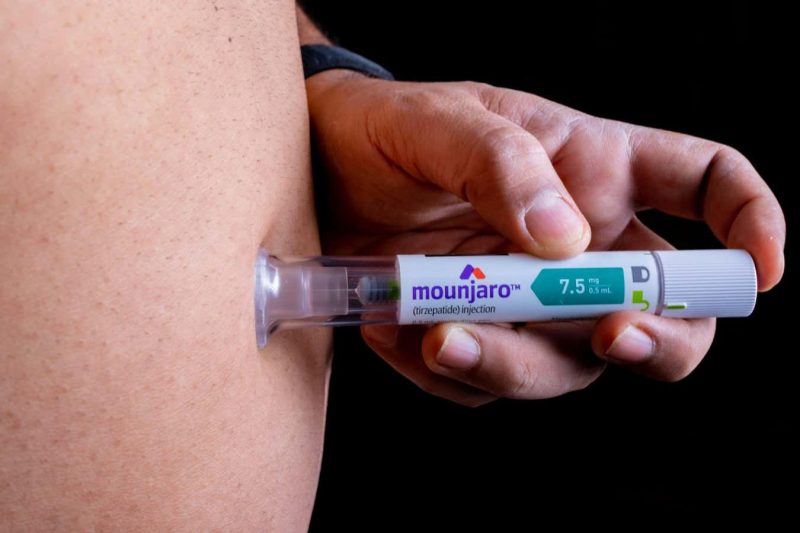
Tirzepatide is now marketed under the brand name Zepbound and is a once-weekly injection that mimics two hormones: GLP-1 and GIP. The combination of these hormones appears to be the key to its effectiveness.
Zepbound has also shown benefits in reducing cholesterol levels, insulin, and blood pressure, along with improving physical mobility.
Considerations and Side Effects
While Zepbound offers promising results, it does come with potential side effects such as nausea, constipation, diarrhoea, and vomiting. There is also a risk of weight regain after discontinuation, a common concern with weight loss medications.
Long-term health risks and potential side effects should be carefully considered before using Zepbound. It is important to note that individuals with specific medical histories should avoid using this medication.
Future Implications
Despite the side effects and potential risks, many believe that the health benefits of Zepbound outweigh its downsides. The approval of Zepbound opens up opportunities for improving the health and quality of life for individuals grappling with obesity.
Conclusion
The approval of Zepbound marks a significant milestone in weight management. However, careful consideration of its side effects and long-term implications is essential for individuals considering this medication for weight loss.