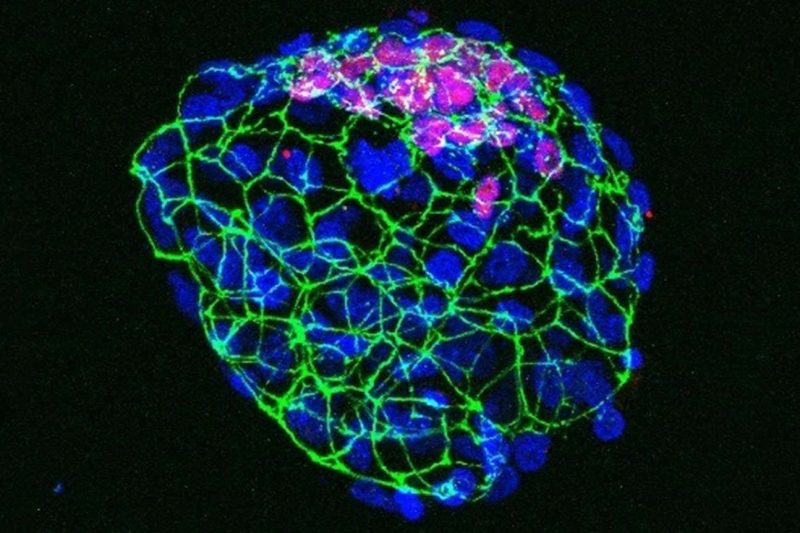
A mouse embryo that has paused its growth resulting from nutrient depletion
Jiajia Ye
The embryos of many species can cease growing when starved of vitamins, solely to restart the method as soon as these are restored – and scientists might have discovered how they do it.
Within the early phases of being pregnant, a fertilised egg turns right into a blastocyst, a tiny cluster of dividing cells. This then implants into the uterine wall, finally differentiating into the assorted organ tissues of a fetus.
When some animals face excessive circumstances, similar to when meals is scarce or when it’s actually chilly, blastocysts pause their development and enter a state of dormancy known as embryonic diapause. This may final for a number of months in some species, with exercise resuming as soon as situations enhance. “It’s a technique to maximise the reproductive course of and thus the variety of younger born and their survival,” says Bruce Murphy on the College of Montreal in Canada, who wasn’t concerned within the analysis.
Now, Jiajia Ye on the Chinese language Academy of Science and his colleagues have uncovered how an embryo can inform when to stall growth.
They put 14 newly pregnant mice in a cage with meals and 11 others in a cage with out meals. After 3.5 days, they discovered that the blastocysts of the well-fed mice developed as ordinary, however these of the starved mice hadn’t implanted within the uterus, indicating embryonic diapause.
When these dormant blastocysts have been then transplanted into the uteruses of well-fed mice, they began rising once more.
In one other a part of the experiment, the researchers grew mouse embryos in petri dishes with completely different vitamins. They discovered that embryonic diapause appears to be attributable to a scarcity of carbohydrates and proteins, whereas embryos uncovered to regular ranges of those vitamins develop as anticipated.
Nearer inspection revealed {that a} sensor known as Gator1 within the blastocysts can detect drops in carbohydrate and protein ranges within the uterus. This then prevents a molecule that controls protein synthesis from being activated, which is critical for blastocyst growth.
When the staff injected the uteruses of pregnant mice that had been disadvantaged of meals with the required carbohydrates and proteins, embryonic development resumed.
With an identical proces anticipated to happen in human embryos, Ye hope these findings may finally be used to enhance fertility therapies. Previous to in-vitro fertilisation (IVF), embryos are typically preserved by freezing, then transplanted right into a uterus. This methodology of preservation is pricey and the embryos don’t at all times survive the thawing course of. The staff has proven it may very well be doable to protect them by way of nutrient depletion, says Ye.
Matters: